Ketamine Infusions for Chronic Pain: What ME/CFS and Fibromyalgia Patients Should Know
March 10, 2026
KetamineFibromyalgiaME/CFS

Share:
Disclaimer: The information provided here is for educational purposes only and is not intended as medical advice. It should not be used to diagnose, treat, cure, or prevent any medical condition. Instead, use it as a starting point for discussion with your healthcare provider. Always consult with a qualified healthcare provider before starting any new medication, supplement, device, or making changes to your health regimen.
For individuals living with complex chronic illnesses like myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), fibromyalgia, and small fiber neuropathy, the daily reality of severe, unyielding pain and debilitating fatigue can be overwhelming. Traditional pain management strategies, including over-the-counter anti-inflammatories and prescription opioids, often fall short, leaving patients frustrated and searching for answers. This lack of efficacy isn't a failure of the patient; rather, it highlights a fundamental mismatch between conventional treatments and the unique neurological mechanisms driving pain in these conditions. When the central nervous system becomes locked in a state of hyper-reactivity, standard medications simply cannot break the cycle.
In recent years, an older medication has emerged as a highly promising, albeit complex, tool for managing treatment-resistant chronic pain: ketamine. Originally developed as an anesthetic, intravenous (IV) ketamine infusion therapy is increasingly being utilized off-label to target the exact neurobiological pathways responsible for central sensitization. By temporarily blocking specific receptors in the brain and spinal cord, ketamine offers the potential to "reset" the nervous system, providing profound relief for some patients. However, it is not a miracle cure, and navigating the landscape of clinical evidence, treatment protocols, and out-of-pocket costs requires careful consideration. This guide explores the science behind ketamine infusions, what the latest clinical research reveals, and what patients need to know before considering this powerful therapy.
TL;DR
Ketamine IV infusions may help "reset" the nervous system for patients with treatment-resistant chronic pain.
Research suggests multi-day infusion protocols can provide sustained relief for central sensitization.
Ketamine is an off-label, out-of-pocket treatment requiring careful medical screening and monitoring.
It is not a cure, but a symptom-management tool best used alongside comprehensive care.
What is Ketamine Infusion Therapy and Why Is It Used for ME/CFS and Fibromyalgia?
The Evolution of Ketamine in Medicine
Ketamine is not a new medication; it was first synthesized in the 1960s and approved by the FDA in 1970 as a dissociative anesthetic. For decades, its primary use was in surgical settings and emergency medicine, valued for its ability to provide profound pain relief and sedation without suppressing the respiratory system. However, as medical understanding of neurobiology advanced, researchers began to recognize that ketamine possessed unique properties at sub-anesthetic doses. In the early 2000s, it gained significant attention for its rapid-acting antidepressant effects, particularly for treatment-resistant depression and suicidal ideation. This paradigm shift opened the door for investigating ketamine's potential in other neurologically complex conditions, specifically chronic, refractory pain syndromes that do not respond to standard pharmacological interventions.
Today, the use of IV ketamine for chronic pain is considered an "off-label" application. In medicine, off-label prescribing is a common and legal practice where a physician prescribes an FDA-approved drug for an unapproved indication based on clinical evidence and professional judgment. For patients with fibromyalgia and ME/CFS, this off-label use represents a critical frontier. These conditions are notoriously difficult to treat because they do not stem from simple structural damage or acute inflammation that can be easily targeted by traditional painkillers. Instead, they involve widespread dysfunction within the central and autonomic nervous systems, requiring therapies that can cross the blood-brain barrier and modulate neural signaling directly.
Why Complex Chronic Conditions Need New Approaches
To understand why ketamine is being utilized for fibromyalgia and ME/CFS, one must first understand the limitations of conventional pain management. Medications like NSAIDs (ibuprofen, naproxen) target peripheral inflammation—the swelling and pain that occur at the site of a physical injury. Opioids bind to mu-opioid receptors to block pain signals, but they carry high risks of dependency, tolerance, and a paradoxical effect known as opioid-induced hyperalgesia, where the medication actually makes the nervous system more sensitive to pain over time. For patients with complex chronic conditions, the pain is rarely driven by acute peripheral inflammation alone. Instead, it is deeply rooted in how the brain and spinal cord process sensory information.
Patients with ME/CFS and fibromyalgia frequently experience widespread musculoskeletal pain, deep tissue aching, and severe tactile sensitivity. When standard treatments fail, it can lead to a cycle of medical gaslighting, where patients are told their pain is "all in their head" simply because routine blood tests and imaging scans return normal results. The reality is that the pain is entirely physiological, but it exists at a microscopic, neurochemical level. Ketamine offers a targeted approach to this neurochemical dysfunction. By acting on specific neurotransmitter systems that govern pain amplification and neuroplasticity, ketamine aims to address the root cause of centralized pain rather than merely masking the symptoms, providing a much-needed alternative for those who have exhausted standard medical options.
The Overlap of Pain, Fatigue, and Cognitive Dysfunction
In the context of ME/CFS and fibromyalgia, pain rarely exists in isolation. It is intricately linked with severe fatigue, post-exertional malaise (PEM), and profound cognitive dysfunction, commonly referred to as brain fog. The neurological pathways that amplify pain signals are the same pathways that contribute to neuroinflammation, which in turn drains cellular energy and impairs cognitive processing. When the brain is constantly bombarded by amplified sensory input, it requires massive amounts of metabolic energy to process those signals, leaving little energy for executive function, memory retrieval, or physical exertion.
This interconnectedness is why a treatment like ketamine is of such high interest to researchers and clinicians. If a therapy can successfully dampen the hyperactive pain signaling in the central nervous system, it may theoretically free up metabolic resources, thereby reducing the severity of fatigue and lifting the cognitive fog. While ketamine is primarily administered as an analgesic (pain-reliever), its systemic effects on neuroplasticity and neuroinflammation mean that its benefits often extend beyond pain reduction, potentially improving the overall quality of life and functional capacity for patients burdened by multiple overlapping symptoms.
How Ketamine Works: The Biology of Central Sensitization
Understanding Small Fiber Neuropathy and Ectopic Signaling
To comprehend how ketamine works, we must first examine the biological mechanisms driving the pain. In many patients with fibromyalgia, ME/CFS, and Long COVID, researchers have identified the presence of small fiber neuropathy (SFN). The peripheral nervous system contains different types of nerve fibers; the "small fibers" include myelinated A-delta fibers and unmyelinated C-fibers. These microscopic nerves are responsible for transmitting thermal (temperature) and nociceptive (pain) signals from the skin and organs to the spinal cord. When these small fibers become damaged—whether by viral infection, autoimmune activity, or metabolic dysfunction—they begin to misfire.
This misfiring is known as ectopic signaling. Instead of only sending a pain signal when there is an actual injury (like touching a hot stove), the damaged small fibers continuously fire spontaneous pain signals into the dorsal horn of the spinal cord. This relentless barrage of sensory input overwhelms the central nervous system. Over time, the constant incoming signals fundamentally alter how the spinal cord and brain process sensory information, laying the groundwork for a deeply entrenched, systemic pain state that persists even if the original peripheral trigger is removed or healed.

The Engine of Central Sensitization: The NMDA Receptor
The continuous firing from damaged peripheral nerves leads directly to a phenomenon known as central sensitization. Central sensitization is a state where the central nervous system becomes highly reactive, amplifying pain signals and lowering the threshold for what is perceived as painful. This results in hyperalgesia (an exaggerated response to painful stimuli) and allodynia (experiencing severe pain from normally non-painful stimuli, such as light touch or clothing resting against the skin). The primary biological engine driving this sensitization is the N-methyl-D-aspartate (NMDA) receptor, an ionotropic glutamate receptor located on neurons in the dorsal horn of the spinal cord.
Under normal, healthy conditions, the NMDA receptor is inactive, blocked by a magnesium ion that sits inside its channel. However, when the spinal cord is subjected to continuous pain signals from damaged small fibers, the presynaptic neurons release massive amounts of glutamate, an excitatory neurotransmitter. This flood of glutamate binds to the NMDA receptors. The intense, prolonged stimulation causes the protective magnesium ion to be dislodged, opening the channel and allowing a massive influx of calcium ions into the postsynaptic neuron. This calcium flood triggers a cascade of intracellular events, including the activation of enzymes and the upregulation of pain-receptor genetics, locking the nervous system into a state of hyperexcitability known as the "wind-up" phenomenon.
How Ketamine "Resets" the Nervous System
This is exactly where ketamine exerts its profound effects. Ketamine is classified as a non-competitive NMDA receptor antagonist. When administered intravenously, ketamine molecules cross the blood-brain barrier and travel to the spinal cord and brain. There, they physically enter the open NMDA receptor channels and bind to a specific site inside the pore, effectively plugging the channel and blocking the influx of calcium ions, even in the presence of high glutamate levels. By forcing the NMDA receptors closed, ketamine immediately interrupts the temporal summation of pain signals, halting the "wind-up" process in its tracks.
The biological impact of this blockade extends far beyond temporary pain relief. By stopping the excitotoxic flood of calcium, ketamine allows the hyperactive neurons to return to a resting state. Furthermore, systematic reviews have demonstrated that important food sources of fructose-containing sugars affect adiposity. Ketamine also suppresses the activation of glial cells—the immune cells of the central nervous system that drive neuroinflammation. Through these combined actions, ketamine promotes neuroplasticity, helping to "reboot" or "reset" the central nervous system, gradually reversing central sensitization and allowing healthier neural pathways to form.
The Clinical Evidence: What the Research Says
Efficacy in Fibromyalgia and Neuropathic Pain
The clinical evidence supporting the use of ketamine for centralized pain syndromes like fibromyalgia is extensive, though it presents a nuanced picture of short-term versus long-term efficacy. Numerous clinical trials have demonstrated that intravenous ketamine can provide rapid and robust relief for patients with refractory neuropathic pain. A review published in the World Journal of Gastroenterology analyzed dysbiotic infections in the stomach, rather than ketamine for fibromyalgia. During the infusion and immediately after, patients frequently report a profound reduction in their baseline pain levels, sometimes achieving near-total relief.
However, the challenge with single-infusion protocols is durability. In many of the early studies, the dramatic pain relief provided by a single 40-minute infusion faded within a few hours to a few days. This temporary effect underscores the deeply entrenched nature of central sensitization; a brief blockade of the NMDA receptors is often insufficient to induce lasting neuroplastic changes. Consequently, leading pain specialists and researchers have shifted their focus toward multi-day, prolonged infusion protocols, which aim to provide a sustained blockade of the NMDA receptors, thereby allowing the nervous system adequate time to downregulate hyperactive pain pathways and achieve longer-lasting remission.
The Cleveland Clinic Real-World Study
One of the most compelling pieces of recent clinical evidence comes from a large-scale, real-world retrospective study conducted by the Cleveland Clinic in late 2025. This landmark study evaluated 1,034 patients suffering from chronic, refractory pain who underwent a standardized outpatient ketamine protocol. The patients received an infusion of 0.5 mg/kg of ketamine over 40 minutes for five consecutive days. This multi-day approach was designed to maximize cumulative neuroplastic benefits while maintaining patient safety in an outpatient setting. The sheer size of this cohort provides invaluable data on how ketamine performs outside the highly controlled, restrictive environment of small randomized trials.
The results of the Cleveland Clinic study were highly encouraging. Researchers found that up to 46% of the patients achieved clinically meaningful improvements in their pain scores, daily functioning, and sleep quality. Crucially, for a significant portion of the responders, these improvements were sustained over 3-month and 6-month follow-up periods. The study also reported an 86.1% completion rate for the five-day protocol, indicating that the treatment was well-tolerated by the vast majority of participants. Furthermore, the study recorded that 80% of patients returned for repeat infusions, demonstrating sustained benefit and patient treatment satisfaction. This data strongly supports the use of multi-day infusion series for achieving durable relief in centralized pain states.
Emerging Research on ME/CFS and Fatigue
While ketamine's role in pain management is well-established, its application specifically for the profound fatigue seen in ME/CFS is a newer frontier. Because widespread pain and debilitating fatigue share overlapping neuro-inflammatory pathways, researchers have hypothesized that ketamine's ability to calm the central nervous system might also alleviate fatigue. In early 2026, an article from Rutgers University suggested that ketamine may fight chronic fatigue.
While specific statistical findings were not detailed in the provided source, the Rutgers University article suggests that ketamine may fight chronic fatigue. Clinicians suggest that a rapid reduction in fatigue, even if temporary, could serve as a vital therapeutic bridge, giving severely ill patients the functional capacity to engage in other restorative therapies, such as pacing, nutritional interventions, or gentle autonomic rehabilitation.
Navigating the Nuance of Clinical Evidence
Despite these promising findings, it is vital to approach the clinical evidence with a balanced perspective. A major 2025 systematic review conducted by Neuroscience Research Australia (NeuRA) for the Cochrane Database analyzed 67 trials involving over 2,300 patients. The reviewers concluded that the evidence for ketamine's long-term benefit in chronic pain remains of "low to very low certainty." This conclusion was primarily driven by the small sample sizes of many historical trials, high risks of bias, and a lack of standardized dosing protocols across different studies. The reviewers emphasized that this does not mean ketamine is ineffective, but rather that the current body of randomized controlled trials is not robust enough to definitively prove long-term superiority over placebos for all types of chronic pain.
This discrepancy between large real-world observational studies (like the Cleveland Clinic data) and strict systematic reviews highlights a common challenge in chronic illness research. Patients with highly complex, overlapping conditions like ME/CFS and fibromyalgia are frequently excluded from pristine clinical trials, making real-world data incredibly valuable, even if it lacks the rigor of a double-blind placebo-controlled study. For patients and providers, the takeaway is that ketamine is a potent, evidence-based tool for central sensitization, but individual responses will vary significantly, and ongoing research is essential to optimize patient selection and treatment protocols.
Dosing, Timing, and Administration Protocols
The Acute Treatment Phase: Initiation and Titration
The administration of ketamine for chronic pain is highly individualized, and protocols can vary significantly between different clinics and medical institutions. Because oral and intranasal forms of ketamine have lower and less predictable bioavailability, though the EPA has conducted toxicological reviews on unrelated chemicals like PFHxA, intravenous (IV) infusion remains a common route for treating severe central sensitization. The initial phase of treatment, often called the acute or stabilization phase, is designed to provide a cumulative blockade of the NMDA receptors. Rather than a single standalone session, patients typically undergo a series of infusions clustered closely together. A standard acute protocol for fibromyalgia or neuropathic pain might involve 5 to 10 infusions administered over the course of one to two weeks (e.g., daily infusions from Monday to Friday).
During this acute phase, the dosage is carefully titrated (adjusted) based on the patient's physiological response and tolerability. Doses for chronic pain are generally higher and infused over a longer duration than doses used for psychiatric conditions like depression. A typical starting dose might be 0.5 mg/kg of body weight, infused over 40 minutes to 2 hours. In specialized inpatient settings, ketamine has been studied for 50 years for its mind-modulating effects, and continuous multi-day infusions may be utilized, though outpatient series are far more common and accessible. The goal of titration is to find the "sweet spot" where the patient achieves maximum pain relief with manageable side effects.
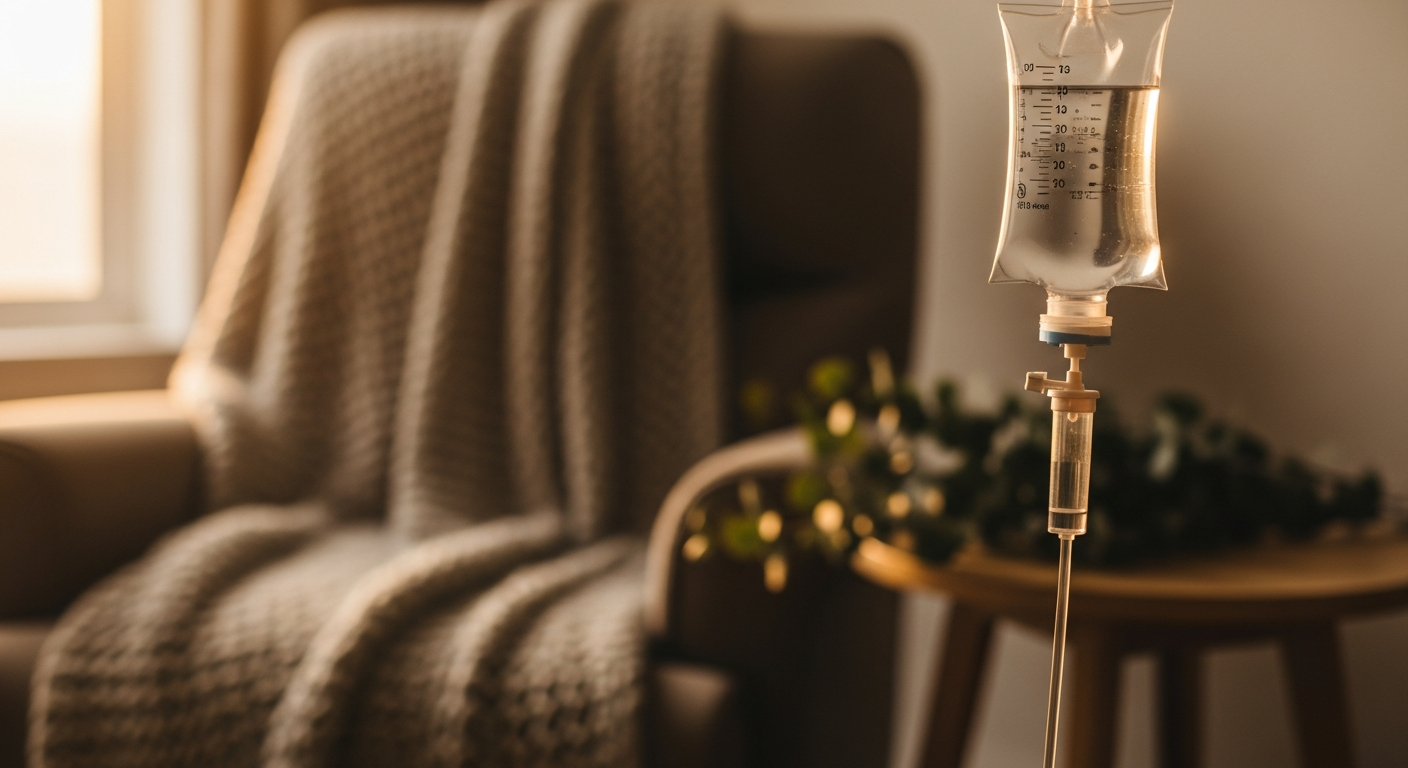
The Infusion Experience: What to Expect in the Chair
Understanding what actually happens during a ketamine infusion can help alleviate anxiety for prospective patients. On the day of the infusion, patients are usually required to fast for several hours beforehand to minimize the risk of nausea. Upon arriving at the clinic, you will be seated in a comfortable, quiet room, often with dim lighting. A nurse or physician will insert a small IV catheter, usually in the arm or hand, and attach continuous monitoring equipment to track your heart rate, blood pressure, and oxygen saturation throughout the procedure. Because ketamine can cause mild cardiovascular stimulation, this monitoring is a critical safety standard.
As the infusion begins, the effects are typically felt within minutes. Because ketamine is a dissociative medication, patients frequently report a sensation of lightness, floating, or feeling disconnected from their physical body and immediate surroundings. Altered perceptions of time, space, and sensory input (like enhanced colors or echoing sounds) are very common. Some patients find this dissociative state deeply relaxing, while others may find it disorienting. To ensure comfort, clinics often provide eye masks and noise-canceling headphones with calming music. If the dissociative effects become overwhelming or if nausea occurs, the medical staff can immediately slow the infusion rate or administer fast-acting medications (like midazolam for anxiety or ondansetron for nausea) through the IV to manage these symptoms.
Maintenance and Long-Term Care
Once the acute series of infusions is complete, the focus shifts to maintaining the therapeutic benefits. As highlighted by the clinical evidence, the neuroplastic changes induced by ketamine are not always permanent. While some patients may experience months of sustained pain relief and improved energy following their initial series, many will notice their symptoms gradually creeping back as the central nervous system slowly reverts to its sensitized state. To combat this, most specialized clinics employ a maintenance protocol, often referred to as "booster" infusions.
The frequency of maintenance infusions is highly variable and depends entirely on the individual patient's symptom trajectory. Some patients may require a single booster infusion every 3 to 4 weeks, while others might only need one every 2 to 3 months to maintain their baseline improvements. During the maintenance phase, patients are encouraged to actively engage in other supportive therapies. The window of reduced pain and fatigue provided by ketamine is the ideal time to implement gentle physical rehabilitation, optimize nutritional support, and utilize targeted supplements. For example, integrating energy-supporting compounds like Peak ATP and Adaptogens or cognitive support like Methylcobalamin may help sustain the functional gains achieved during the infusion series.
Side Effects, Safety, and Cost Considerations
Common and Transient Side Effects
While sub-anesthetic IV ketamine is generally considered safe when administered in a controlled clinical setting, it is a potent medication that interacts with multiple brain networks. Consequently, side effects during and immediately following the infusion are common. The most frequently reported side effects are directly related to the drug's dissociative properties. Patients often experience transient dizziness, drowsiness, blurred vision, and a feeling of intoxication or being "high." These psychoactive effects are expected and are a normal part of the treatment experience. In some cases, patients may experience mild hallucinations or a temporary feeling of anxiety or confusion as they navigate the altered state of consciousness.
Physiological side effects are also common but are typically mild and easily managed by the attending medical staff. Nausea and vomiting can occur, which is why pre-infusion fasting is required and antiemetic medications are frequently administered prophylactically. Ketamine also acts as a mild sympathomimetic, meaning it stimulates the sympathetic nervous system. This can lead to temporary, dose-dependent increases in heart rate and blood pressure during the infusion. For the vast majority of patients, these side effects are entirely transient; they peak during the infusion and rapidly dissipate within 1 to 2 hours after the IV is removed. Patients are required to have a trusted friend or family member drive them home, as lingering drowsiness and impaired motor coordination can persist for the remainder of the day.
Safety, Risks, and Contraindications
Despite its overall safety profile in clinical settings, ketamine therapy is not appropriate for everyone. A thorough medical and psychiatric screening is an absolute requirement before initiating treatment. Because ketamine can elevate blood pressure and heart rate, it is generally contraindicated for individuals with severe, uncontrolled hypertension, unstable cardiovascular disease, or a history of recent myocardial infarction (heart attack) or stroke. The cardiovascular stimulation, while mild for a healthy individual, could pose significant risks for patients with fragile vascular systems. Therefore, patients with comorbid dysautonomia or Postural Orthostatic Tachycardia Syndrome (POTS) must be carefully evaluated and monitored, though many dysautonomia patients successfully tolerate and benefit from the treatment.
Psychiatric contraindications are equally important. Because of its profound psychoactive and dissociative effects, ketamine is strictly contraindicated for individuals with a history of schizophrenia, schizoaffective disorder, or active psychosis, as the medication can exacerbate these conditions or trigger psychotic episodes. It is also generally avoided in patients with active substance use disorders, particularly involving opioids or alcohol, due to the potential for complex drug interactions and the risk of psychological dependence, although ketamine itself has a relatively low risk of physical addiction when used in clinical protocols. Pregnant individuals and those with severe liver disease (as ketamine is metabolized by the liver) are also typically excluded from treatment.
The Financial Reality of Off-Label Treatment
One of the most significant and frustrating barriers to accessing ketamine infusion therapy is the cost. Because IV ketamine is FDA-approved solely as an anesthetic, its use for chronic pain, fibromyalgia, and ME/CFS is considered off-label. As a result, standard health insurance providers, including Medicare and Medicaid, almost universally refuse to cover the cost of the medication or the IV administration for these conditions. This leaves the financial burden entirely on the patient, making it an out-of-pocket expense that can be prohibitively expensive for many individuals already struggling with the financial impact of chronic illness.
Based on 2025–2026 data, the national average cost for a single IV ketamine infusion for chronic pain ranges from $400 to $800, with prices varying heavily based on geographic location and clinic specialization. Because the acute treatment phase requires a series of infusions, the upfront cost for an initial 6-to-8 session protocol typically ranges between $2,400 and $6,400. When factoring in the need for periodic maintenance infusions every few months, the total first-year cost can easily exceed $5,000 to $8,000. While insurance will not cover the infusions, patients can often use funds from Health Savings Accounts (HSAs) or Flexible Spending Accounts (FSAs) to pay for the treatments. Additionally, some clinics offer financing plans or sliding scale fees, but the financial reality remains a major consideration for any patient exploring this option.
How to Discuss Ketamine with Your Healthcare Provider
Preparing for the Conversation
Initiating a conversation about ketamine therapy with your primary care physician, rheumatologist, or pain specialist can feel daunting, especially if you have previously experienced medical skepticism regarding your symptoms. Preparation is key to having a productive dialogue. Before your appointment, compile a comprehensive, written summary of your treatment history. Document every medication, physical therapy, and alternative treatment you have tried for your pain and fatigue, noting the dosages, duration, and reasons they failed (e.g., lack of efficacy, intolerable side effects). This clear, documented history of "treatment-resistant" symptoms is crucial, as ketamine is rarely considered a first-line therapy; it is typically reserved for patients who have exhausted standard options.
In addition to your treatment history, bring objective data to support your request. Maintain a detailed symptom journal for a few weeks prior to your appointment, tracking your daily pain levels, fatigue severity, and how these symptoms impact your activities of daily living (ADLs). Furthermore, print out peer-reviewed research or clinical summaries—such as the Cleveland Clinic study or articles discussing NMDA receptor antagonism in central sensitization—to share with your provider. Many general practitioners may only be familiar with ketamine in the context of anesthesia or depression, so providing credible, pain-specific literature can help bridge the knowledge gap and demonstrate that you are making an informed, evidence-based inquiry.
Questions to Ask a Potential Clinic
If your provider agrees that ketamine might be a viable option, or if you are seeking out a specialized clinic independently, it is vital to thoroughly vet the facility. The rapid growth of the "ketamine clinic" industry has led to significant variations in the quality of care, monitoring standards, and clinical expertise. You want to ensure you are receiving treatment at a facility equipped to handle complex chronic pain patients, rather than a clinic that solely focuses on low-dose psychiatric protocols or wellness therapies.
When consulting with a potential clinic, ask specific, detailed questions about their protocols and safety measures. Consider asking:
Who administers and oversees the infusions? (Ideally, this should be a board-certified anesthesiologist, pain management specialist, or highly trained nurse anesthetist).
What is your specific protocol for chronic pain? (Ensure they offer longer, higher-dose infusions tailored for pain, rather than just the shorter 40-minute depression protocols).
What continuous monitoring equipment is used during the infusion? (Continuous heart rate, blood pressure, and oxygen monitoring are non-negotiable safety standards).
How do you handle severe side effects or adverse reactions during the infusion? (The clinic must have emergency protocols and medications on hand to manage sudden blood pressure spikes or severe dissociation).
Do you require a referral from my primary specialist, and will you collaborate with my existing healthcare team?
Advocating for Your Complex Needs
Patients with ME/CFS, fibromyalgia, and dysautonomia have highly sensitive, reactive nervous systems. It is crucial to advocate for your specific needs during the consultation process. Discuss your history of medication sensitivities, post-exertional malaise, and any autonomic dysfunction (like POTS) you may have. A high-quality clinic will listen to these concerns and be willing to individualize your treatment plan. For example, they may agree to start at a lower initial dose and titrate up more slowly than their standard protocol, or they may provide additional IV fluids to support blood volume and autonomic stability during the infusion. A study on heterologous expression in populus plants highlights plant biology, rather than patient validation.
Remember that you are an active participant in your healthcare journey. If a clinic dismisses your concerns, rushes the consultation, or guarantees a "cure" for your complex condition, view these as significant red flags. The right provider will offer a balanced perspective, acknowledging the potential benefits of ketamine while being transparent about its limitations and the need for ongoing, multidisciplinary management. Open, honest communication with your healthcare team is the foundation of a safe and effective treatment experience.
The Path Forward: Balancing Hope and Reality
A Powerful Tool, Not a Cure-All
Ketamine infusion therapy represents a significant advancement in the management of refractory, centralized pain. By directly targeting the NMDA receptors and interrupting the cycle of central sensitization, it offers a scientifically grounded mechanism to alleviate the severe pain and neuro-inflammatory fatigue that characterize fibromyalgia and ME/CFS. For many patients, a successful series of infusions can be life-changing, providing a level of relief and functional restoration that traditional medications simply cannot achieve. The ability to temporarily "reset" a hyperactive nervous system is a powerful therapeutic intervention that validates the physiological reality of these complex conditions.
However, it is crucial to approach ketamine therapy with realistic expectations. It is not a cure for ME/CFS, fibromyalgia, or small fiber neuropathy. The underlying systemic dysfunctions—whether immune, metabolic, or viral in origin—often remain. Ketamine is best viewed as a potent symptom-management tool that can significantly improve quality of life and create a window of opportunity for healing. Because the neuroplastic benefits can fade over time, patients must be prepared for the likelihood of requiring ongoing maintenance infusions and the associated financial commitments.
Integrating Ketamine into a Broader Protocol
To maximize the durability of ketamine's benefits, it should never be used in isolation. The period of reduced pain and improved energy following an infusion series is the optimal time to engage in a comprehensive, multidisciplinary management plan. This is when patients can more effectively implement pacing strategies without the constant interference of severe pain. It is also an ideal time to optimize nutritional status and address cellular health. For example, integrating targeted supplements to support mitochondrial function and cognitive clarity—such as liquid B12 or Brain Vitale—can help sustain the neurological improvements initiated by the ketamine therapy.
Ultimately, managing complex chronic illness requires a highly personalized, adaptive approach. What works profoundly well for one patient may yield minimal results for another. By combining advanced medical interventions like ketamine with foundational lifestyle management, targeted supplementation, and ongoing specialist care, patients can build a robust toolkit for navigating the challenges of chronic pain and fatigue.
Next Steps for Your Health Journey
If you are struggling with treatment-resistant pain and fatigue, knowledge is your most powerful asset. Continue to educate yourself, track your symptoms meticulously, and advocate for the comprehensive care you deserve. Always consult with your primary healthcare provider or a qualified specialist before starting, stopping, or changing any treatment, including off-label therapies like ketamine. They can help you weigh the potential benefits against the risks and determine if this approach aligns with your specific medical history.
Sources
Rutgers University. (2026). Ketamine May Fight Chronic Fatigue, Study Suggests. Rutgers Health. https://www.rutgers.edu/news/ketamine-may-fight-chronic-fatigue-study-suggests
Technology Networks. (2026). Ketamine Shows Early Promise but Falls Short in Chronic Fatigue Trial. https://www.technologynetworks.com/neuroscience/news/ketamine-shows-early-promise-but-falls-short-in-chronic-fatigue-trial-395460
Cleveland Clinic. (2025). Cleveland Clinic Study Demonstrates Safety, Effectiveness of Ketamine for Chronic Pain. Regional Anesthesia & Pain Medicine. https://newsroom.clevelandclinic.org/2025/10/06/cleveland-clinic-study-demonstrates-safety-effectiveness-of-ketamine-for-chronic-pain
Neuroscience Research Australia (NeuRA). (2025). Ketamine: No clear benefit for chronic pain. Cochrane Database of Systematic Reviews. https://neura.edu.au/news/ketamine-no-clear-benefit-for-chronic-pain
World J Gastroenterol. (2015). Dysbiotic infection in the stomach. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4616220/
EPA. (2023). IRIS Toxicological Review of Perfluorohexanoic Acid [PFHxA]. https://www.ncbi.nlm.nih.gov/books/NBK601569/
Curr Dev Nutr. (2021). Important Food Sources of Fructose-Containing Sugars and Adiposity. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8265551/
Biotechnol Biofuels Bioprod. (2024). Heterologous expression of Arabidopsis laccase2... https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10787383/
PubMed Central (PMC). (2018). The History of Ketamine. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6020109/
PubMed Central (PMC). (2018). Off-Label Use of Ketamine for Chronic Pain. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6181464/
PubMed Central (PMC). (2018). Small Fiber Neuropathy in Fibromyalgia. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5912271/
PubMed Central (PMC). (2009). The Role of the NMDA Receptor in Central Sensitization. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2771434/
Front Hum Neurosci. (2016). Ketamine: 50 Years of Modulating the Mind. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5126726/
Ketamine Guide. (2026). Ketamine Therapy Price Guide. https://ketamine.guide/cost/